Fabrication of a novel magnetic CdS nanorod/NiFe2O4/NaX zeolite nanocomposite with enhanced sonocatalytic performance in the degradation of organic dyes
Abstract
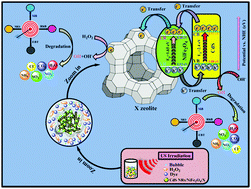
In the present study, a novel magnetic CdS nanorods (NRs)/NiFe2O4/NaX zeolite sonocatalyst nanocomposite was successfully synthesized for the first time by a simple ultrasound-assisted solvothermal method. Several techniques, including FTIR, FE-SEM, EDAX, EDAX elemental dot-mapping, TEM, AFM, XRD, VSM, UV-Vis DRS, and N2-BET, were utilized to characterize the as-fabricated catalyst. Then, the CdS nanorods/NiFe2O4/NaX catalyst nanocomposite was used for the degradation of various organic dyes, such as methylene blue (MB), eriochrome black T (EBT), rhodamine B (RhB), and methyl orange (MO), in aqueous solutions under ultrasound irradiation, and the results were obtained via UV-Vis absorption analysis. The sonodegradation processes of the dye contaminants were carried out in the presence of H2O2 as an oxidizing agent. Moreover, the effects of different experimental parameters, namely irradiation time, process type, catalyst dosage, initial dye concentration, H2O2 concentration, scavenger type, and catalyst reusability, on the degradation reactions of MB, EBT, RhB, and MO were investigated. Furthermore, to study the sonodegradation reactions of dyes, the first-order kinetic model was employed. Subsequently, the rate constant (kapp) and half-life (t1/2) for the degradation of MB in the US/H2O2/CdS NRs/NiFe2O4/NaX system were measured as 0.0538 min−1 and 12.881 min, respectively. In addition, the acquired information reveals the prominent role of ˙OH, known as a radical scavenger, derived from H2O2 in the entire sonodegradation reaction.


 Please wait while we load your content...
Please wait while we load your content...