Unusual effects of the bulky 1-norbornyl group in cobalt carbonyl chemistry: low-energy structures with agostic hydrogen atoms†
Abstract
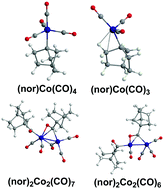
The 1-norbornyl (nor) ligand is known experimentally to form stable transition metal alkyl derivatives through direct metal–carbon bond formation. This appears to be related to its steric bulk and inaccessibility towards β-hydrogen elimination, as exemplified by the tetraalkyls (nor)4M, some of which are very stable. In this connection we have used density functional theory and the DLPNO-CCSD(T) method to investigate the 1-norbornylcobalt carbonyl derivatives (nor)Co(CO)n (n = 4, 3, 2, 1) and (nor)2Co2(CO)n (n = 7, 6, 5). Low-energy structures of the unsaturated systems (nor)Co(CO)n (n = 3, 2) and (nor)2Co2(CO)n (n = 6, 5) are found to have agostic hydrogen atoms from a CH2 group adjacent to the Co–C bond. Such agostic hydrogen atoms form a C–H–Co bridge with a bonding Co–H distance less than ∼2 Å. In such structures unsaturation is relieved by donation of an additional two electrons from the C–H bond of this norbornyl CH2 group. In addition, structures in which carbonyl migration from cobalt to carbon has occurred to form acyl norCO ligands are among the lowest energy structures. The resulting acyl carbonyl groups of the norCO ligands serve as spacers between the bulky 1-norbornyl ligand and the cobalt carbonyl moiety. Furthermore, such neutral norCO acyl ligands can either be one-electron donors to a cobalt atom, bonding solely through the carbonyl carbon, or three-electron donor η2-μ-norCO groups bridging a central Co2 unit through both the acyl carbon and oxygen atoms. The strengths of the agostic C–H–Co interactions have been characterized by their reduced density gradient (RDG) values.


 Please wait while we load your content...
Please wait while we load your content...
