The impact of a hygroscopic chitosan coating on the controlled release behaviour of zinc hydroxide nitrate–sodium dodecylsulphate–imidacloprid nanocomposites
Abstract
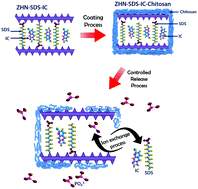
Imidacloprid (IC) is a neutral charge insecticide that is commonly used in paddy cultivation areas to kill invasive insects. However, the uncontrolled usage of IC may cause serious problems for non-target organisms and the environment. This work focusses on the potential of using chitosan to develop an efficient insecticide that could minimise the environmental risk by controlling the release of IC into the environment. The powder X-ray diffraction (PXRD) and Fourier transform infrared (FTIR) analysis of the chitosan coated zinc hydroxide nitrate–sodium dodecylsulphate–imidacloprid (ZHN–SDS–IC-Chi) nanocomposite confirmed that the chitosan coating process did not interfere with the types of ions that were intercalated in the interlayer gallery of the nanocomposite. The release study performed in aqueous solutions of Na3PO4, Na2SO4 and NaCl revealed that the slowest release was observed when using NaCl as the release media. The release of IC was governed by the pseudo second order kinetic model, which described the release mechanism of the IC as being via dissolution and ion exchange. Even though the overall findings showed that the physicochemical properties of the ZHN–SDS–IC and ZHN–SDS–IC-Chi were not too different, the presence of chitosan was found to be beneficial for prolonging the release process. The synthesised ZHN–SDS–IC-Chi nanocomposite will hopefully be used as a safer insecticide in paddy cultivation and will fulfil both economic and ecological demands.


 Please wait while we load your content...
Please wait while we load your content...