Design and identification of poly(vinyl chloride)/layered double hydroxide@MnO2 nanocomposite films and evaluation of the methyl orange uptake: linear and non-linear isotherm and kinetic adsorption models†
Abstract
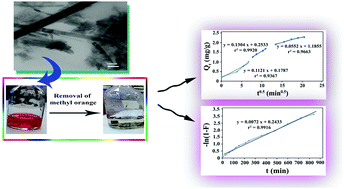
In this work, an adsorbent based on poly(vinyl chloride) (PVC) for the removal of methyl orange (MO) dye is proposed. In this regard, a hybrid composed of modified layered double hydroxide (M-LDH) and α-MnO2 nano-rods was used as a nano-filler and a series of PVC based nanocomposite films were prepared using solution casting. The prepared PVC/M-LDH@MnO2 nanocomposites containing 5, 10, and 15 wt% of the nano-filler were characterized via several techniques. Then, they were used for the removal of MO from aqueous solution. The 10 wt% nanocomposite film showed the best performance, so it was used to attain the optimum conditions. Two isotherm models including Langmuir and Freundlich were used for fitting the adsorption data. Also, the kinetics of adsorption was investigated using pseudo-1st order and pseudo-2nd order models. Both linear and non-linear forms were applied in each case and the best model was selected based on the correlation coefficients (r2) and some error functions. In addition, both intraparticle and film diffusion were probable mechanisms in the adsorption process.


 Please wait while we load your content...
Please wait while we load your content...