Heterogeneous assembly of Pt-clusters on hierarchically structured CoOx@SnPd2@SnO2 quaternary nanocatalysts manifesting oxygen reduction reaction performance†
Abstract
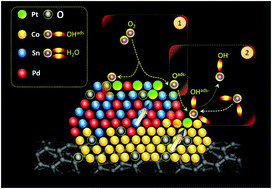
The rational design and synthesis of heterogeneous interfaces with tailored structural and functional properties are highly sought to realize green energy technologies. In the present study, quaternary metallic heterogeneous nanocatalysts (NCs) consisting of Pt-cluster decorated CoO2–SnPd2–SnO2 hierarchical structures (namely CSPP) are proposed with improved heteroatomic interactions for an oxygen reduction reaction (ORR) in alkaline medium (0.1 M KOH). The CSPP NCs have been synthesized with different Pt contents (1, 2 and 14 wt%) by using a wet chemical reduction method on a carbon nanotube (CNT) support with simultaneous heterogeneous and homogenous nucleation. Of special relevance, the mass activity (MA) of CSPP-1 (∼1.0 wt% Pt) and CSPP-2 (∼2.0 wt% Pt) NCs is 2146.2 mA mg−1 and 1555.7 mA mg−1, which is ∼32 and ∼23-fold increased, respectively, as compared to that of the commercial J.M.-Pt/C catalyst (20 wt% Pt) at 0.85 V vs. RHE. Through intensive analysis of microscopy and spectroscopy results, we demonstrated that such enhanced ORR activities for the ultra-low dosage of Pt are mainly dominated by incorporation of Pt atoms into the defect sites of the Co–Sn–Pd surface. These Pt-atoms lower the adsorption strength for oxygenated species via electron confinement from adjacent sites, resulting in enhanced splitting and relocation kinetics of subsequent oxygen molecules on the NC surface and thus ORR performance.


 Please wait while we load your content...
Please wait while we load your content...