Preparation of a g-C3N4/Co3O4/Ag2O ternary heterojunction nanocomposite and its photocatalytic activity and mechanism†
Abstract
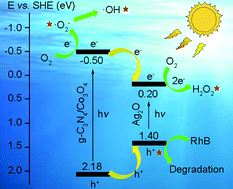
Development of semiconductor catalysts is an attractive way to solve energy shortage and pollution problems. In this work, a kind of ternary heterojunction nanocomposite g-C3N4/Co3O4/Ag2O was successfully prepared, whose morphology, structure and composition were characterized by TEM, EDS, XRD and XPS analysis. Photocatalytic experiments prove that g-C3N4/Co3O4/Ag2O shows good photodegradation efficiency (98.09%) for RhB under visible light. The free radical capture experiment indicates that the hole, superoxide radical and hydroxyl radical all contribute to the photocatalytic process. In addition, it is found that introducing Ag2O into the composite can efficiently promote the formation of H2O2via the reduction of O2. An electrochemical oxygen reduction reaction (ORR) study further confirms that the value of electron transfer during the ORR is 2.07 for g-C3N4/Co3O4/Ag2O, indicating the promoted two-electron O2 reduction reaction to H2O2. Based on the above results and energy band structures of semiconductors, a possible photocatalysis mechanism is proposed.


 Please wait while we load your content...
Please wait while we load your content...