Electrochemical detection of 2-nitroaniline at a novel sphere-like Co2SnO4 modified glassy carbon electrode
Abstract
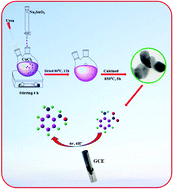
A novel electrochemical sensor, based on a Co2SnO4 (CoSnO) modified glassy carbon electrode (GCE), has been successfully developed for the determination of 2-nitroaniline (2-NA). The CoSnO was synthesized by a simple co-precipitation method and characterized by FE-SEM, HR-TEM, EDX, FT-IR and XRD. The electrochemical performance of the CoSnO modified GCE towards the reduction of 2-NA was investigated by cyclic voltammetry (CV) and differential pulse voltammetry (DPV). CoSnO/GCE exhibits strong electrocatalytic activity towards the reduction of 2-NA. The electrochemical studies show that the CoSnO/GCE has superior electrocatalytic activity, whereas the unmodified GCE has lower reduction potential response. Under the optimal conditions, the proposed sensor responses were in a wide linear range from 0.04 μM to 856.14 μM with a low detection limit of 0.062 μM. Moreover, the as-synthesised CoSnO is studied for selectivity, stability, reproducibility and repeatability. In the presence of interfering species like nitro groups and inorganic species (cationic and anionic), the reduction peak current response doesn’t cause any variation in results and discloses good selectivity towards the detection of 2-NA. Finally, it is made clear that the proposed sensor CoSnO/GCE exhibits excellent electrochemical behaviour for electrochemical determination towards reduction of 2-NA.


 Please wait while we load your content...
Please wait while we load your content...