Hydrogenation of CO2 to LPG over CuZnZr/MeSAPO-34 catalysts†
Abstract
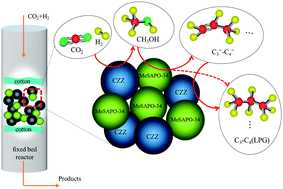
The utilization of CO2 to synthesize environmentally benign liquid fuels offers a solution to replacing depleting petroleum resources. Herein, a ternary CuZnZr (CZZ) metal oxide catalyst and a SAPO-34 zeolite were synthesized by co-precipitation and hydrothermal synthesis, respectively. Different metals were impregnated into the latter to obtain MeSAPO-34 (Me = Mn, Zn and Zr). A granule mixture of CZZ and MeSAPO-34 components (CZZ/MeSAPO-34 catalyst) was then effectively utilized in a tandem catalytic process for one-step CO2 hydrogenation to liquefied petroleum gas (LPG). The CZZ/MeSAPO-34 catalysts were characterized by using XRD, H2-TPR, BET, SEM-EDS and NH3-TPD techniques. SEM-EDS and XRD results indicated that an appropriate amount of Zr metal loading induced minimum zeolite framework collapse compared to a similar amount of Mn and Zn, which was more favorable for higher activity. In addition, NH3-TPD results revealed that the acidity of SAPO-34 could be altered after impregnation with different metals in different quantities. Tuning the acid density and strength, together with adjusting the CZZ to MeSAPO-34 weight ratio, had a collectively critical effect on LPG selectivity. An effective hydrogenation microenvironment which favors lower alkane formation (C3–C4) was enhanced after the acidity of the molecular sieve was tuned. LPG selectivity could reach 86% over the CZZ/5% ZrSAPO-34 catalyst at 2 MPa, 350 °C, a W/F ratio of 6, a H2/CO2 ratio of 3 and a weight ratio of 1.


 Please wait while we load your content...
Please wait while we load your content...