Photoelectrocatalytic properties and mechanism of rhodamine B degradation using a graphene oxide/Ag3PO4/Ni film electrode†
Abstract
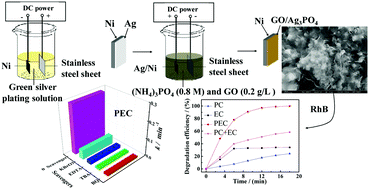
Graphene oxide (GO)/Ag3PO4/Ni composite film electrodes were prepared by composite electrodeposition using a mixture of ammonium phosphate and GO aqueous suspension as the electrolyte. The morphology, crystallinity, and optical characteristics of the composite films were analyzed. The photoelectrocatalytic (PEC) degradation activity of the film electrodes towards rhodamine B (RhB) was determined by applying an anodic bias under visible-light irradiation. The PEC mechanism of the GO/Ag3PO4/Ni film electrodes was explored by investigating the photo-generated charge behavior and the active species of the PEC reaction. The results showed that the GO/Ag3PO4/Ni composite films prepared by the optimum process consisted of GO coated on Ag3PO4 nanospheres with a diameter of about 100 nm. Under the optimum anodic bias, their PEC degradation efficiency toward RhB was 1.74 times that of an Ag3PO4/Ni film and 4.47 times higher than their photocatalytic (PC) efficiency without an applied bias. Importantly, the GO/Ag3PO4/Ni composite films exhibited an obvious photoelectric synergistic effect and excellent PEC stability; at an anodic bias of 0.1 V, its their PEC stability was about twice their PC stability. The excellent charge conduction properties of GO and its ability to separate photogenerated electrons and holes more effectively under an anode bias and with an increase in the concentration of ˙RhB+ were the main reasons for the high PEC activity of the composite films.


 Please wait while we load your content...
Please wait while we load your content...