Interplay between porous texture and surface-active sites for efficient oxygen reduction reactions in N-inherited carbon†
Abstract
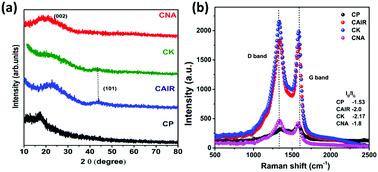
Nitrogen-doped porous carbon materials have excellent oxygen reduction reaction (ORR) activities due to their synergistic effects caused by the electron-accepting ability of the adjacent sp2 bonded carbon atoms leading to the redistribution of charge density. However, the nitrogen doping in the carbon matrix usually involves a tedious chemical synthesis process with toxic compounds. So, the synthesis of nitrogen-doped carbon from nitrogen-containing biomass with simpler carbonization methods would be advantageous. Herein, we report a facile synthesis of nitrogen-enriched porous activated carbon from mandarin peel biomass. The prepared carbon possesses uniform pore distribution, and a highly mesoporous and defective nature with enriched nitrogen and oxygen species over the carbon surface. The excellent ORR activity is confirmed by the rotating ring electrode (RDE) measurements. The onset potentials of ORR are 0.83, 0.81, and 0.87 V vs. RHE and the half-wave potentials are 0.7, 0.71, and 0.75 V vs. RHE for open-air-, KOH- and NaOH-treated samples, respectively. All the samples follow the conventional four-electron transfer process with good stability in an alkaline medium. We have observed a correlation between the onset potential and nitrogen content of carbon. Moreover, the kinetic current increases with an increase of specific surface area due to higher electrochemically active surface sites. The honeycomb-like morphology allows the electrolyte to penetrate through the porous network and enhance the ORR activity. This study reveals a trade-off between nitrogen content, porous texture, and surface-active sites.
- This article is part of the themed collection: NJC Editorial Board web collection


 Please wait while we load your content...
Please wait while we load your content...