A biodegradable nanodrug of molybdenum silicide for photothermal oncotherapy†
Abstract
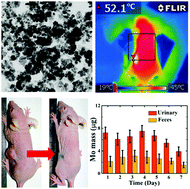
Photoactive nanomaterial-mediated photothermal therapy is a promising cancer treatment method. However, its development is hindered by the in vivo toxic potential of nanomaterials, which may stay in the body for a long time. One of the ideal strategies to overcome such a predicament is exploiting biodegradable photoactive substances for photothermal therapy. In this work, MoSi2 nanoparticles were found to be excellent materials for near-infrared (NIR) activation and showed a broad photoabsorption band in the NIR range and resultant NIR-triggered photothermal effect. On the basis of its unique photoactive properties, MoSi2 was applied as a phototherapeutic agent in this work for photothermal oncotherapy. The in vitro cell tests and in vivo antitumor studies on mice both manifested a prominent antitumor outcome, as evidenced by the efficient cancer cell killing and solid tumor removal. More importantly, MoSi2 has been proved as a body-clearable nanodrug that can be degraded in vivo and excreted via feces and urine, thus lowering the long-term toxic potential. In addition, MoSi2 has excellent biosafety, which has been proven by the low levels of cytotoxicity, hematotoxicity and tissue toxicity. Consequently, MoSi2 as a nanodrug is an ideal candidate for photothermal cancer treatment with adequate biosecurity.


 Please wait while we load your content...
Please wait while we load your content...