Fusion of thienyl into the backbone of electron acceptor in organic photovoltaic heterojunctions: a comparative study of BTPT-4F and BTPTT-4F†
Abstract
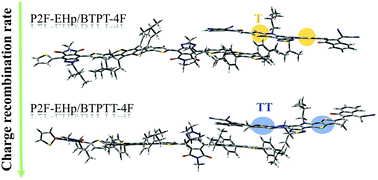
Molecular engineering of electron donors and acceptors for organic bulk heterojunction solar cells is critical to develop novel materials with improved power conversion efficiency. Here, the non-fullerene acceptors (NFAs) BTPT-4F and BTPTT-4F combined with an electron donor, P2F-EHp, were taken as a representative system to theoretically study the effects of the fusion of thienyl into the backbone of NFAs. On the basis of extensive quantum chemistry calculations, it was found that fusing thienyl into the backbone elevated the highest occupied molecular orbital (HOMO) energy, reduced the gap between the HOMO and the lowest unoccupied molecular orbital, decreased the dipole moment, enhanced the light absorbance and generated red-shifts in the absorption bands. The data for the P2F-EHp/BTPT-4F and P2F-EHp/BTPTT-4F complexes indicate that fusing thienyl into the backbone of the electron acceptor modifies the excitation character of the donor/acceptor complexes, increases the charge transfer (CT) excitation energy and the number of local excitation and CT hybridized low-lying excited states, amplifies the CT rate, reduces the exciton dissociation rate, and dramatically suppresses the charge recombination (CR) rate, leading to better performance of BTPTT-4F. However, the poorer performance of BTPT- 4F can mainly be attributed to its larger dipole moment and faster CR rate.


 Please wait while we load your content...
Please wait while we load your content...