Selective oxidation of aniline into azoxybenzene catalyzed by Nb-peroxo@iron oxides at room temperature
Abstract
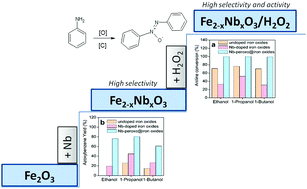
Oxidation of aniline into valuable products such as azoxybenzene, azobenzene, and nitrobenzene has been a great challenge due to the difficulty of controlling the selectivity of heterogeneous catalysts to form the desired products. In this work, an active and selective heterogeneous catalyst for the oxidation of aniline into azoxybenzene was designed by doping iron oxide supports with niobium and immobilizing Nb-peroxo groups on their surfaces through H2O2 treatment. The Nb-peroxo groups on the iron oxide support were essential to catalyze the conversion of aniline into products while the niobium favored the formation of azoxybenzene. Among the alcoholic solvents studied, 1-propanol was the most suitable to get high conversion rates of aniline and high selectivity to azoxybenzene. At 3 h reaction time, the Nb-peroxo@iron oxides led to oxidation of aniline with a conversion rate of 99.6% and 83.7% selectivity to azoxybenzene at room temperature. Since the selective and catalytic oxidation of aniline is very important for the synthesis of intermediates and precursors to various industrially valuable products, the results shown herein may contribute to the development of most stable, selective, and active catalysts under mild reaction conditions.


 Please wait while we load your content...
Please wait while we load your content...