Development and functionalization of magnetic nanoparticles as stable and reusable catalysts for triacetin synthesis†
Abstract
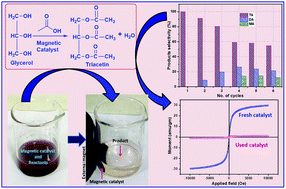
The application of magnetic nanoparticles as an adsorbent and a catalyst support in solving the environmental issues has recently received great attention due to their unique physical and chemical properties, such as high surface area, ease of anchoring the functional groups, lower processing cost, high stability and ease of separation under an external magnetic field. In the present study, a magnetic catalyst was prepared by embedding magnetite (Fe3O4) within the silica shell on which sulfate ions were anchored (Fe3O4@SiO2@SO42−). The catalyst was successfully employed for glycerol esterification with acetic acid to obtain 100% selectivity towards triacetin within 45 min of reaction duration at 80 °C. The catalyst was successfully isolated from the reaction medium magnetically and reused for six consecutive reaction cycles while retaining 90% glycerol conversion levels and 50% triacetin selectivity in the reaction cycles.


 Please wait while we load your content...
Please wait while we load your content...