Investigation of phosphogypsum valorization by the integration of the Merseburg method
Abstract
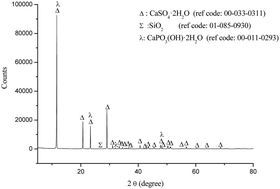
The proposal of a method for converting the phosphogypsum (PG) waste remains the focus of many researchers since its quantity, growing considerably, is causing serious environmental problems. In the present study, PG was converted into lithium sulfate and calcium sulfate solids in a continuous process through the integration of the Merseburg method. Firstly, the ammono-carbonation of PG was studied by varying the reaction time in a microwave apparatus. Based on the characterization results, 2 hours of reaction led to a PG's conversion yield of 60% to the calcite phase. After transforming the ammonium sulfate to lithium sulfate and studying its thermal stability, the investigation of the electric conduction of the latter compound using impedance spectroscopy showed interesting results making it a good candidate for applications in battery devices. The calcium sulfate, produced after CaCO3 processing, was of high purity. This was verified by scanning electron microscopy/energy dispersive X-Ray spectroscopy (SEM/EDX) which showed a rod morphology predominantly composed of Ca, S and O with a very low Cl content.


 Please wait while we load your content...
Please wait while we load your content...