Cobalt-based layered double hydroxides revisited: evidence for oxidizing radical generation†
Abstract
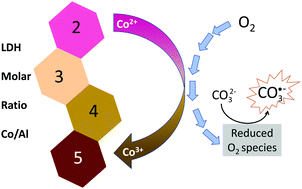
Layered double hydroxides (LDHs) containing transition metal elements such as cobalt have been studied mainly for applications in electrochemical devices and as catalysts. Fundamental studies regarding Co-based LDHs are required and are attractive owing to the complexity of cobalt chemistry. In this work, a series of LDH materials intercalated with carbonate and comprising different cobalt and aluminium molar ratios (R = 2, 3, 4, and 5) were synthesized using a coprecipitation method and characterized by X-ray diffraction (XRD), UV/Vis electronic spectroscopy, infrared (IR) and Raman spectroscopies, X-ray photoelectron spectroscopy (XPS), thermal analysis coupled with mass spectrometry (MS), scanning electron microscopy (SEM) and electron paramagnetic resonance (EPR). XRD patterns pointed out single phase materials, while XPS spectroscopic data indicated the presence of trivalent cobalt. EPR spectral profiles show three components of g-factor, indicating that Co(II) locates in a rhombic symmetry (x ≠ y ≠ z), associated with Jahn–Teller octahedral distortion. The signal intensity decreases with the increase of Co/Al ratio, suggesting the presence of a cation with diamagnetic characteristics, possibly Co(III) (3d6 electronic system with low-spin) and/or dipole–dipole interaction. Furthermore, EPR spectra of all the synthesized materials display a fine signal with a g value equal to 2.060 and a 16.9 Gauss width, possibly related to the presence of carbonate radical ions, since this signal is not observed in the spectra of cobalt-based LDH analogues intercalated with chloride or nitrate ions. Considering the high oxidizing power of carbonate radicals, materials prepared under the experimental conditions reported in this work have promising potential for applications requiring an oxidative environment such as the radical polymerization of organic compounds and degradation of pollutants.
- This article is part of the themed collection: Sustainability from intercalation compounds


 Please wait while we load your content...
Please wait while we load your content...