Ligand rigidity and electronic effect on the complexation of hexavalent plutonyl with three dicarboxylic acids: a combined spectrophotometric and computational study†
Abstract
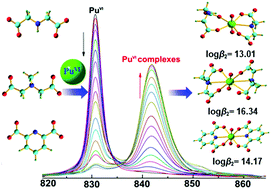
Uncovering the complexation properties of actinides with common organic ligands in aqueous solution is of significant importance to understand the fundamental coordination chemistry of actinides as well as to develop more efficient extractants for actinides separation in advanced nuclear fuel cycles. Herein, the complexation behaviour of hexavalent plutonyl (Pu(VI) or PuO22+) with three dicarboxylic acids, iminodiacetic acid (IDA), N-methyl-iminodiacetic acid (MIDA) and dipicolinic acid (DPA), was studied through both experimental and theoretical approaches. In 1.0 M NaClO4 medium, successive formation of 1 : 1 and 1 : 2 (metal/ligand ratio) complexes of Pu(VI) with IDA and MIDA were identified and their stability constants were determined and compared with those of DPA. The complexation strength varies for the complexes of Pu(VI) with IDA, MIDA and DPA. For the 1 : 1 complexes, the stability constants of Pu(VI) with the three ligands follow the order of IDA < MIDA < DPA, while those of the 1 : 2 complexes follow the order of IDA < DPA < MIDA. The complexation trends are validated by DFT calculations, which suggest that the complexation strength of IDA/MIDA/DPA with Pu(VI) is governed by both the electronic effect (electron withdrawn group or electron donating group) and the rigidity of the ligands. Furthermore, the symmetric features of the Pu(VI) complexes with the three ligands were illustrated by analyzing the optical characteristics of the complexes and DFT calculation. The results provide new insights into the intrinsic mechanism of hexavalent actinide complexation with tridentate dicarboxylic acids in aqueous solutions.


 Please wait while we load your content...
Please wait while we load your content...