Fragmentation method reveals a wide spectrum of intramolecular hydrogen bond energies in antioxidant natural products†
Abstract
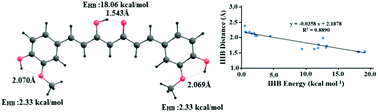
Intramolecular hydrogen bonds (IHBs) have been reported to have a pronounced effect on the biological activity of naturally occurring compounds. Despite this, the direct estimation of IHB energies in natural products has not been attempted. In this work, we report the IHB energies in some well-known antioxidant natural products using a molecular tailoring approach (MTA)-based method. Our estimates reveal a wide spectrum of IHB strengths in these compounds with hydrogen bond (H-bond) energies ranging from ∼0.6 to 19 kcal mol−1 at the B3LYPD3/6-311G(d,p) and ∼0.5 to 18 kcal mol−1 at the MP2/6-311G(d,p) level of theory. The strong IHBs are seen to be resonance-assisted. The bifurcated O–H⋯O![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C hydrogen bonds have moderate energies, viz. 6 to 7 kcal mol−1. Other types of weak IHBs such as O–H⋯OCH3, O–H⋯OH, and O–H⋯O(ring) have energies in the range from ∼0.50 to ∼2.8 kcal mol−1. The trends in the IHB energies are in accordance with the rank-ordering based on the respective H-bond distances, H-bond angles and molecular electron density values at the (3, −1) bond critical points.
C hydrogen bonds have moderate energies, viz. 6 to 7 kcal mol−1. Other types of weak IHBs such as O–H⋯OCH3, O–H⋯OH, and O–H⋯O(ring) have energies in the range from ∼0.50 to ∼2.8 kcal mol−1. The trends in the IHB energies are in accordance with the rank-ordering based on the respective H-bond distances, H-bond angles and molecular electron density values at the (3, −1) bond critical points.


 Please wait while we load your content...
Please wait while we load your content...