A highly active Pd/H-ZSM-5 catalyst in lean methane combustion prepared via a sol–gel method and treated by reduction–oxidation†
Abstract
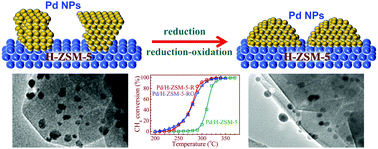
Catalytic combustion is now considered to be an effective measure in the mitigation of anthropogenic methane emission contributing to the greenhouse effect, which demands appropriate catalysts of high activity at low temperatures and with high stability at elevated temperatures. Herein, a series of Pd/H-ZSM-5 catalysts were prepared via a sol–gel method and treated by reduction and oxidation; the effect of Pd particle size and redox treatment on the catalytic activity of Pd/H-ZSM-5 in lean methane combustion was investigated. The results indicate that the catalytic activity of Pd/H-ZSM-5 varies according to a volcano-shape trend with an increase in the Pd particle size from 3.5 to 10.1 nm; the Pd/H-ZSM-5 catalyst with a Pd particle size of around 5.2 nm exhibits the highest activity in lean methane oxidation. Moreover, the catalytic activity of Pd/H-ZSM-5 can be greatly enhanced through the reduction–oxidation treatment, which may reconstruct the palladium particles on the H-ZSM-5 surface and strengthen the interaction between the Pd species and the acid sites on the H-ZSM-5 support. The Pd/H-ZSM-5 catalyst after the reduction and oxidation treatment (with a Pd particle size of 5.2 nm) exhibits excellent activity in lean methane combustion, giving complete methane oxidation to carbon dioxide at as low as 300 °C. This work should be of great benefit for clarifying the relationship between the structure and performance for the acid zeolite supported Pd catalysts and for designing efficient catalysts in the lean methane combustion at low temperature.


 Please wait while we load your content...
Please wait while we load your content...