The synthesis and characterization of an Fe3O4@TiO2–Ni/rGO magnetic photocatalyst
Abstract
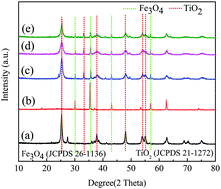
In this study, Fe3O4@TiO2–Ni core/shell particles linked to graphene oxide was fabricated via sol–gel and hydrothermal methods as a photocatalyst. The composition, crystalline structure, morphology, and surface physicochemical properties were characterized via X-ray diffraction (XRD), scanning electron microscopy (SEM) coupled with X-ray photoelectron spectroscopy (XPS), transmission electron microscopy (TEM), UV diffuse reflectance spectroscopy (UV-DRS), photoluminescence (PL), and nitrogen adsorption–desorption measurements. The results show that the magnetic photocatalyst consists of Fe3O4@TiO2–Ni active particles and rGO carriers. The active particles have a double-shell core–shell structure with a size of about 400 nm, and are supported on the rGO lamellae. The Fe3O4@TiO2–Ni/rGO microspheres can be easily collected by applying an external magnetic field due to the magnetic property of the core Fe3O4 particles. TiO2 doping with a small amount of metal Ni and rGO can significantly improve the catalytic effect of the magnetic photocatalyst, and rGO can also significantly improve the adsorption of pollutants by the magnetic photocatalyst. Under visible light irradiation, Fe3O4@TiO2–Ni/rGO was able to degrade 95.4% of rhodamine B (RhB) within 140 min, and this ability remained at 90.15% after 7 times of repetitive use under the same conditions.


 Please wait while we load your content...
Please wait while we load your content...