Effect of pre-shearing treatment on the molecular structure, fibrillogenesis behavior and gel properties of collagen
Abstract
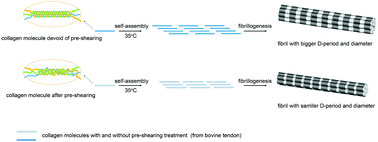
Shear treatment is always employed in the extraction, preparation and research of collagen materials. This study is aimed at investigating the effect of the pre-shearing treatment of neutral bovine tendon collagen solution on the molecular structure, assembly behavior and gel properties of collagen. Rheological and assembly degree (AD) tests showed that with the increase in the shear rate or the extension of shear time during the pre-shearing treatment, the equilibrium moduli and AD of collagen gels that assemble from the pretreated samples gradually decreased. Scanning electron microscopy (SEM) results indicated that with the deepening of the pre-shearing treatment (increased shear rate or shear time), the diameter of the resultant collagen fibers gradually decreased, and their pore size and porosity increased as a whole. The viscosity test of the collagen solution obtained after pre-shearing revealed that the viscosities of the samples were lower than that of the sample devoid of pre-shearing. Sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS–PAGE) and circular dichroism (CD) analysis manifested that the molecular weights and CD characteristic peaks of the collagen molecules remained unchanged in the sheared solution. However, with the deepening of the pre-shearing treatment, the proportion of α chains in the SDS–PAGE image increased, while the total content of β and γ chains decreased; the triple-helix structure of collagen in the CD spectrum decreased, and the random coiled structure increased. Moreover, transmission electron microscopy (TEM) displayed that the D-periods of the fibers assembled from the sheared samples were also decreased. A mechanism for the effect of the pre-shearing treatment of the collagen solution on the molecular structure, assembly behavior and gel properties of collagen has been proposed. The findings in this work may provide new understanding and theory on the physical modification of proteins and the construction of new materials from polymer systems that are aggregated or assembled based on weak intermolecular interactions.


 Please wait while we load your content...
Please wait while we load your content...