Structure and electrochemical properties of hierarchically porous carbon nanomaterials derived from hybrid ZIF-8/ZIF-67 bi-MOF coated cyclomatrix poly(organophosphazene) nanospheres†
Abstract
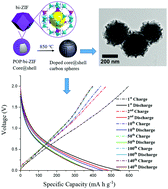
Hybrid bi-ZIF nanocrystals consisting of ZIF-8/ZIF-67 were synthesised in the presence of cyclomatrix poly(organophosphazene) (POP) nanospheres and formed POP/bi-ZIF core@shell nanospheres. POP/bi-ZIF showed excellent thermal stability up to 478 °C, with well-preserved core@shell structures during carbonization at 850 °C. The resultant core@shell carbon nanospheres exhibited hierarchically mesoporous structures. The porous carbon core was derived from the carbonised covalent inorganic–organic polyphosphazene framework, containing in situ doped heteroatoms such as N, P, S and O; the shell structure was derived from bi-ZIF containing up to 40% Zn and Co elements. The bi-ZIF derived carbon shell showed a BET surface area of 1347.76 m2 g−1 and a Langmuir surface area of 1882.71 m2 g−1, and the total BET surface area of the core@shell structure reached 1025.00 m2 g−1. When applied as an anode material in lithium ion batteries, the core@shell carbon structure exhibited a charge capacity of 595 mA h g−1 with a discharge capacity of 546 mA h g−1 and maintained a reversible charge/discharge capacity of 400 mA h g−1 after 140 cycles, which is higher than the theoretical capacity of a graphite anode. A good cycling stability with 83% capacity retention in the C-rate tests was achieved. This work provides a facile and scalable method to produce mesoporous carbon nanostructures with in situ doped metal elements and heteroatoms, which benefits the high rate electrochemical properties of lithium ion batteries.


 Please wait while we load your content...
Please wait while we load your content...