Intercalation chemistry and thermal characteristics of layered double hydroxides possessing organic phosphonates and sulfonates†
Abstract
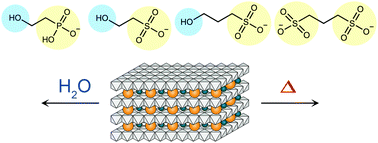
Intercalation of 2-hydroxyethanesulfonate (2-heS) and 3-hydroxy-1-propanesulfonate (3-hpS) into layered Mg–Al double hydroxides (Mg–Al LDHs) allows delamination of layers in water and enables the synthesis of high-surface-area oxides through non-oxidative thermal decomposition. To investigate the roles of anionic heads and hydroxyl tails in these organic sulfonates, 2-hydroxyethanephosphonate (2-heP) and 1,3-propanedisulfonate (1,3-pDS) were tested as analogs of hydroxyalkylsulfonates, and the thermal characteristics of the resultant LDHs were investigated. Exchange of interlayer CO32− with 2-heP proceeds similarly to 2-heS, causing the same degree of interlayer expansion. However, the resultant LDH resists swelling and delamination in water, demonstrating the distinct property enabled by the –SO3− group. Although LDH possessing 1,3-pDS intercalants seems to resist delamination because of the lack of hydroxyl groups, its thermal characteristics resemble those of its 3-hpS counterpart, demonstrating the crucial role of alkyl sulfonate groups to allow the formation of enhanced microporous structures through non-oxidative thermal treatments. Our results show further that even a partial exchange of CO32− by 3-hpS, corresponding to 0.05 equivalents relative to the exchangeable Al sites, triggers unique thermal characteristics observed for LDH-sulfonates.
- This article is part of the themed collection: Sustainability from intercalation compounds


 Please wait while we load your content...
Please wait while we load your content...