Chitosan and pectin core–shell beads encapsulating metformin–clay intercalation compounds for controlled delivery†
Abstract
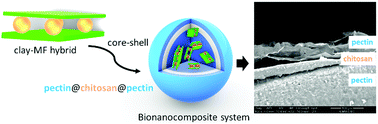
This work explores the development of bionanocomposite systems for controlled drug delivery of metformin, the most extensively used oral drug for treatment of type 2 diabetes, also now proposed for treatment of various types of cancer. These systems involve the incorporation of metformin–clay intercalation compounds into a biopolymer matrix, chitosan or pectin, that it is further covered with one or two more biopolymer coatings to ensure stability in the stomach (pectin coating) as well as mucoadhesive properties (chitosan) to attain controlled release of metformin in the intestinal tract. Thus, intercalation compounds involving the ion-exchange of interlayer cations of a natural Wyoming montmorillonite (Cloisite®Na) and a synthetic hectorite (LAPONITE® XLG) were entrapped in chitosan or pectin matrices, producing bionanocomposite beads that were coated with a pectin layer in the first case, or with a chitosan layer and a second external layer of pectin in the second case, to produce core–shell beads that were tested in the controlled release of metformin. The produced beads were submitted to water stability and in vitro release tests simulating the changes of pH along the gastro-intestinal tract with the aim to establish the performance of each type of core–shell bead as a delivery system. It is expected to profit from the stability provided by pectin to reduce the delivery in the stomach, to improve the residence time of the system in the intestinal tract using the mucoadhesive properties of the chitosan, and to use the drug intercalated in the clay as a reservoir from which it can be slowly released in the intestine, and so the combined action of the three components affords a controlled delivery system for the oral administration of metformin.
- This article is part of the themed collection: Sustainability from intercalation compounds


 Please wait while we load your content...
Please wait while we load your content...