New technique for elaboration and characterization of a high voltage spinel LiCo2O4 cathode and theoretical investigation
Abstract
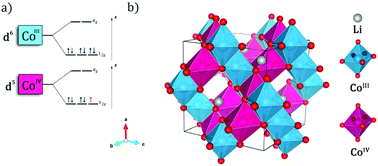
For the first time, the nanoparticles of LiCo2O4 spinel were elaborated at low temperature by a new process using non-standard Moroccan metallurgical grade reagents. Thermogravimetric analysis (TGA) of the as prepared powders was carried out for the determination of decomposition temperature. X-ray diffraction (XRD) patterns confirmed the formation of LiCo2O4 at 400 °C, which has a cubic spinel structure with the space group (Fd3m). The crystallite size and lattice parameters were found to be 19 nm and 8.153 Å, respectively, which illustrate the transition of the spinel to the nanocomposite HT-LiCoO2–Co3O4–CoO at 900 °C. The FTIR (Fourier Transform Infrared) spectra showed three fundamental absorption bands (v1, v2 and v3), which are attributed to the stretching vibrations of octahedral and tetrahedral sites, respectively. Particle size, morphology and chemical composition of spinel LiCo2O4 are evaluated using transmission electron microscopy (TEM) coupled with EDS. The EDS spectrum has confirmed the purity of the compound by the presence of Co and O. In addition, first-principles calculations were performed on the spinel material using the Full-Potential Linear Augmented Plane Wave method. The corresponding electronic structure is studied and the Li average voltage is calculated using the GGA+U method with the present work self-consistently calculated Ueff values.


 Please wait while we load your content...
Please wait while we load your content...