Modification of a H-terminated silicon surface by organic sulfide molecules: the mechanism and origin of reactivity†
Abstract
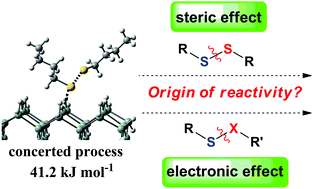
The design of silicon-chalcogenide-based materials is an important topic for functionalized silicon materials, but the mechanism of this process is not so clear yet. In the present work, mechanistic studies were performed on the modification of the H-terminated Si(100) surface by many organic sulfide molecules by using the density functional theory method. Calculations indicate that the mechanism involves two steps: (1) a concerted rather than a stepwise Si–S bond formation and S–S bond dissociation process and (2) an H abstraction process from the direction along rather than perpendicular to the SiH2–Si–SiH2 skeleton. The first step is the rate-determining step for all the reactions considered. For the reactions of disulfide molecules (RSSR), the steric effect of the R group is the main origin of the different reactivity, while the electronic effect of the R group has just a slight effect on the reaction's reactivity. In the reactions of sulfide molecules (RSXR′, X = S, P, Si, O, N, C), for substrates containing X elements in the same period of the periodic table, the activation barrier increases along with the decrease of the atomic number. Charges on the S atom and dissociation energies of the S–X bonds have a great impact on the reactivity of these reactions.


 Please wait while we load your content...
Please wait while we load your content...