Synthesis and investigation of the photophysical, electrochemical and theoretical properties of phenazine–amine based cyan blue-red fluorescent materials for organic electronics†
Abstract
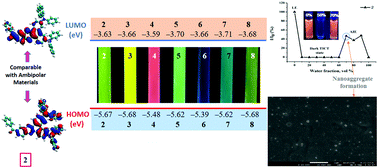
To understand the structure–property relationship, we have designed and synthesized seven novel donor–acceptor based phenazine–amines, 2–8, by the Buchwald–Hartwig coupling amination reaction in moderate yield. The synthesized molecules were fully characterized and the influence of modulating the donor and phenazine core on the optoelectrochemical properties was studied. The absorption spectra of 2–8 display intramolecular charge transfer (ICT) transitions in the range of 431–501 nm. Dyes 2–8 exhibit positive solvatofluorochromism and emit in the cyan blue-red region with emission maxima at 498–641 nm on excitation at their respective ICT maxima in toluene, chloroform, DCM and neat solid film. The photophysical studies of the dyes in different THF/water mixtures revealed aggregation induced emission (AIE) features in dyes 2, 3 and 5. The HOMO and LUMO energy levels were found to be in the ranges of −5.39 to −5.68 eV and −3.59 to −3.71 eV, respectively, with electrochemical band gaps within the range 1.73–2.04 eV. Theoretical studies of the molecules were also carried out by using TD-DFT calculations. The comparable electrochemical energy levels to ambipolar materials, solid state luminescence with AIE characteristics and good thermal stability of the synthesized dyes signify their potential to be used as solid state emitters and ambipolar materials in optoelectronics.


 Please wait while we load your content...
Please wait while we load your content...