Synthetic saponite clays as promising solids for lanthanide ion recovery†
Abstract
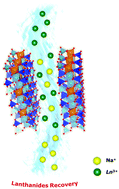
Extraction of lanthanide ions with different ionic radius (La3+, Gd3+ and Lu3+) from aqueous solutions was carried out by using two synthetic saponite clays. The clays, prepared through a simple and low-cost hydrothermal approach, showed different particle size and cation-exchange capacity (CEC) and were characterized in order to evaluate the influence of their physico-chemical properties on the uptake process. Metal uptake tests, performed in pure water and simulated freshwater and seawater solutions, demonstrated that synthetic saponites have a general good sorption capacity with high lanthanide capture after a few hours of contact. The solid samples showed a higher selectivity for Lu3+, due to its small ionic radius.
- This article is part of the themed collection: Sustainability from intercalation compounds


 Please wait while we load your content...
Please wait while we load your content...