Ultrasound-assisted synthesis of rGO/Sb4O5Cl2/Sb2S3 for a high photo-catalytic rate†
Abstract
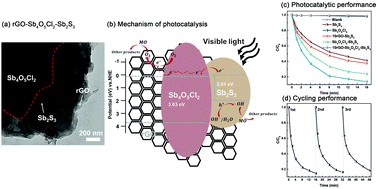
The present study focuses on the synthesis and characterization of a ternary photo-catalyst: rGO–Sb4O5Cl2–Sb2S3. A series of rGO–Sb4O5Cl2–Sb2S3 samples were synthesized by the ultrasonic-assisted chemical co-precipitation method and the photo-catalytic capability was evaluated by the photo-degradation of methyl orange (MO) under visible light irradiation. The photo-catalysis rate of rGO–Sb4O5Cl2–Sb2S3 was higher than those of Sb2S3, 10rGO–Sb2S3, Sb4O5Cl2 and Sb4O5Cl2–Sb2S3. The as-prepared material (30rGO–Sb4O5Cl2–Sb2S3, 5 mg) had the highest photo-degradation effect on MO (50 mL, 20 mg L−1), and 87.4% MO degraded within 16 min. The cycling test has proven that the produced rGO–Sb4O5Cl2–Sb2S3 has anti-photoerosion ability. The morphology, structure and opto-electronic properties have been characterized as well. Thus, the mechanism has been discussed: the extraordinary photo-degradation effect on MO results from the synergetic effect of ultrathin rGO sheets and Sb4O5Cl2 grains. Sb4O5Cl2 was the photo-generated charge carrier migration medium and rGO would enhance the catalytic efficiency because of the ultra-large specific surface area and numerous surface active sites.


 Please wait while we load your content...
Please wait while we load your content...