Unconventional DNA-relevant π-stacked hydrogen bonded arrays involving supramolecular guest benzoate dimers and cooperative anion–π/π–π/π–anion contacts in coordination compounds of Co(ii) and Zn(ii) phenanthroline: experimental and theoretical studies†
Abstract
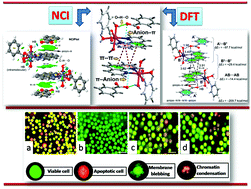
Two new coordination compounds, viz., [Co(phen)(bz)(H2O)3]·bz (1) and [Zn(phen)2(H2O)Cl]·Cl·bzH·2H2O (2) (phen = 1,10-phenanthroline, bz = benzoate, bzH = benzoic acid) were synthesized from purely aqueous media, and characterized using elemental analysis, spectroscopic (IR, UV-Vis-NIR) and single crystal X-ray diffraction techniques. In the crystal structures of 1 and 2, supramolecular networks have been built involving the association of O–H⋯O, C–H⋯O, and π–π stacking interactions. The enclathration of the guest bzH molecule in the host monomers of Zn(II) brings rigidity to the structure of compound 2. Theoretical investigations have been carried out to rationalize interesting supramolecular assemblies that include cooperative anion–π/π–π/π–anion in 1 and DNA-relevant HB-array⋯π/π–π/π–HB-array interactions in 2, which have been characterized using NCI plots and Bader's theory of “atoms-in-molecules”. The complexes significantly inhibit cell viability by inducing apoptotic cell death in a Dalton's lymphoma (DL) cell line with negligible cytotoxicity in normal cells. A molecular docking study and pharmacophore features reveal that compounds 1 and 2 interact and are accommodated well in the active site of anti-apoptotic proteins. Antimicrobial studies against a few pathogenic organisms suggest that the compounds are active against Gram-positive and Gram-negative bacteria and have relatively better antibacterial activity in comparison to phen.


 Please wait while we load your content...
Please wait while we load your content...