Ultrafine PdAg nanoparticles immobilized on nitrogen-doped carbon/cerium oxide for superior dehydrogenation of formic acid†
Abstract
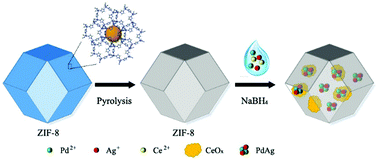
The development of safe and efficient hydrogen storage/release systems is critical for achievement of a fuel-cell-based hydrogen economy. In this work, ultrafine PdAg NPs with the size of 2.5 nm were successfully immobilized on cerium oxide/nitrogen-doped carbon (CeOx–NPC). By virtue of strong metal/support interactions, the obtained PdAg/CeOx–NPC exhibits excellent catalytic activity in FA decomposition at room temperature and the turnover frequency (TOF) reaches a high value of 875 h−1. The remarkable catalytic property of PdAg/CeOx–NPC results from the small and highly dispersed PdAg NPs and the synergistic effect of the bimetallic system. Also, the strong metal–support interaction efficiently improves the catalytic properties of the bimetallic catalytic system.


 Please wait while we load your content...
Please wait while we load your content...