Multi-responsive drug delivery nanoplatform for tumor-targeted synergistic photothermal/dynamic therapy and chemotherapy
Abstract
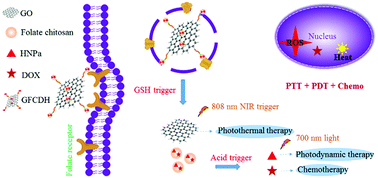
To overcome the serious adverse reactions, multidrug resistance and non-specificity towards tumor tissues of conventional chemotherapy drugs in cancer therapy, and the low efficiency of single photodynamic therapy owing to the hypoxic environment inside tumors, we have prepared a “from one to all” multi-mode therapy integrated nanoplatform. GFCDH nanoparticles with fluorescence imaging function were prepared based on tumor-targeting folate-conjugated chitosan loaded with doxorubicin (DOX) and photosensitizer HNPa (FCDH) as the core, and cystine-functionalized graphene oxide as a vehicle. The on–off drug control was activated by stimulation of the endogenous tumor microenvironment to enhance the intracellular multi-responsive drug delivery for tumor-targeted synergistic photothermal therapy, photodynamic therapy and chemotherapy. The drug-loading capacity of DOX and HNPa in GFCDH nanoparticles was up to 5.24% and 5.58%, respectively, and the loaded drugs could be efficiently released by acid stimulation, glutathione (GSH) reduction and near-infrared light induction. In vitro, 87% of the loaded HNPa was released within 72 hours by GSH/acid stimulation, simulating the tumor microenvironment, compared with 20% at a physiological pH of 7.4. Furthermore, it was shown that irradiation with 808 nm light induced increased drug release. GFCDH nanoparticles possessed good photothermal effect and 1O2 quantum yield (ΦΔ = 42.1%) using methylene blue (ΦΔ = 49.1%) as a reference. An MTT assay showed a higher cancer cell toxicity for GFCDH treatment with 808 nm and 700 nm light irradiation compared with single wavelength light irradiation. All the results reveal the promise for applications of this smart strategy to overcome existing barriers in chemotherapy and photodynamic therapy for enhanced anticancer therapy.


 Please wait while we load your content...
Please wait while we load your content...