Janus coordination polymer derived PdO/ZnO nanoribbons for efficient 4-nitrophenol reduction†
Abstract
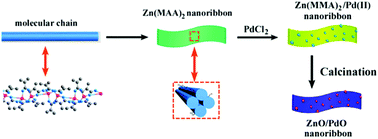
We designed a facile and green self-assembly route for the large-scale production of bimetallic coordination polymer–Zn(MAA)2/Pd(II) nanoribbons by employing two terminal units with distinct hard–soft properties of the smallest semi-rigid methacrylate anion (MAA−) to combine with two different metal ions. The COO− (chemically hard base) served as the initial unit, which was applied to coordinate with the Zn2+ ions for constructing the host network of this Janus CPs by facilely adjusting the proportion of ethanol/water to control the degree of solvation. After soaking in an ethanol solution of PdCl2, the alkene units (chemically soft base) act as secondary donors to load the soft noble metal Pd2+ ions and form Zn(MAA)2/Pd(II) nanoribbons, which are ideal precursors of ZnO/PdO nanoribbons. By pyrolyzing the dual metal ion equipped Zn(MAA)2/Pd(II) precursors, ZnO nanoribbon supported PdO nanoparticles (NPs) with a well-defined structure were facilely fabricated on a large scale. Furthermore, the as-obtained ZnO/PdO nanoribbons exhibit efficient catalytic performance for the reduction of 4-NP, which can convert 99.3% of the substrates within 6 min. This superior catalytic performance may be attributed to the well-dispersed PdO nanoparticles as well as the absence of additional agents introduced a green synthetic method.


 Please wait while we load your content...
Please wait while we load your content...