Chemical bowling-assisted synthesis of Fe3O4@starch-derived carbon composites as anode materials with superior cycling stability for lithium-ion batteries†
Abstract
Fe3O4@starch-derived carbon composites (Fe3O4@C-SD composites) were produced via chemical bowling, an economic and a scalable method, and a subsequent calcination with starch as the carbon resource and iron(III) nitrate as the iron resource. The Fe3O4@C-SD composites present an alveolate-like structure, with ultrafine Fe3O4 nanoparticles on the carbon framework. The Fe3O4@C-SD1 composite delivers a larger specific surface area of ∼132.5 m2 g−1, which can supply more active sites for lithium storage and benefit lithium flux across the interface. The interior hollow structure provides a void space for the volume change of Fe3O4 particles, thus ensuring the integrity of the electrode materials. When used as an anode material, it shows great potential in lithium storage. The specific capacity of the Fe3O4@C-SD1 electrode could reach 581.5 mA h g−1 after 1000 cycles at a current density of 1 A g−1, which indicated that the electrode exhibited an excellent cycling stability during the charge/discharge process. The synergistic effects between the carbon matrix and the Fe3O4 nanoparticles in the Fe3O4@C-SD1 composite led to the superior lithium storage performance. This above-mentioned synthetic strategy is simple, economical, scalable and controllable, thereby providing an approach for the synthesis of other composites with similar structure.
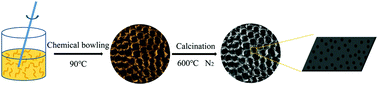


 Please wait while we load your content...
Please wait while we load your content...