Computational prediction of interaction and pharmacokinetics profile study for polyamino-polycarboxylic ligands on binding with human serum albumin†
Abstract
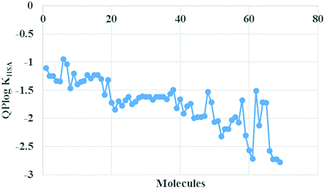
Human serum albumin (HSA) is one of the most abundant plasma proteins available in blood and responsible for transport of fatty acids, drugs and metabolites at its two primary binding sites (site 1 and site 2). The interaction of drug molecules with HSA is important and attention-grabbing in the field of science, chemistry and clinical medicine since it affects drug stability and toxicity during therapeutic processes. Hence, the detailed investigation of HSA–drug interactions is required to understand the pharmacodynamics and pharmacokinetics profile of a drug. A drug molecule with effective pharmacological properties can be designed by studying the HSA binding, which acts as a reservoir for a long duration of action and ultimately affects the ADMET (absorption, distribution, metabolism, excretion and toxicity) properties of the drug molecules. Here in the present work a computational investigation including a binding analysis and interaction study of polyamino-polycarboxylic ligands with HSA was conducted. The in silico analysis has been implemented at HSA drug binding sites, site 1 and site 2, via docking studies, prime-MM-GBSA analysis (molecular mechanics energies with generalized Born and surface area continuum solvation), and multiple linear regression analysis with ADMET descriptors and quantitative estimation of log KHSA for the respective molecules. These descriptors described the relevant pharmacokinetics and pharmacodynamics of these ligands for gadolinium-based MRI agents (GBCA). Finally, a complete picture and correlation tell about the primary aspect for the selection of vehicles for magnetic resonance and lanthanide ion-based optical imaging probes.


 Please wait while we load your content...
Please wait while we load your content...