Synthesis of 1,6-disubstituted pyrene-based conjugated microporous polymers for reversible adsorption and fluorescence sensing of iodine†
Abstract
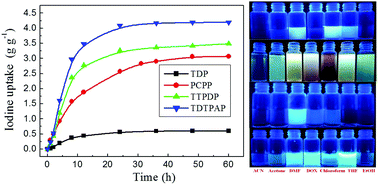
Here we present detailed evidence of highly efficient iodine capture and sensing in 1,6-disubstituted pyrene-based fluorescent conjugated microporous polymers, which were synthesized by a Sonogashira–Hagihara polycondensation reaction (TDP), trimerization reaction of a bicycano compound (CPP) catalyzed using trifluoromethanesulfonic acid (PCPP), and Friedel–Crafts reaction catalyzed with CH3SO3H (TTPDP and TDTPAP), respectively. TDP, PCPP, TTPDP, and TDTPAP have specific surface areas of 261.9, 43.0, 187.5, and 695.2 m2 g−1, and display reversible guest uptake values of 0.61, 3.07, 3.49, and 4.19 g g−1 in iodine vapor, respectively. The four CMPs exhibit high sensitivity and selectivity to iodine via fluorescence quenching. Furthermore, PCPP exhibited extremely high detection sensitivity to I2 with a KSV of 1.40 × 105 L mol−1 and a detection limit of 3.14 × 10−13 mol L−1. To the best of our knowledge, it displays the highest reported KSV value and the lowest detection limit value to iodine to date.


 Please wait while we load your content...
Please wait while we load your content...