Hierarchical construction and characterization of lanthanum molybdate nanospheres as an unassailable electrode material for electrocatalytic sensing of the antibiotic drug nitrofurantoin
Abstract
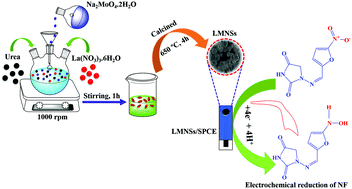
In this work, lanthanum molybdate nanospheres (LMNSs) were prepared by employing a co-precipitation methodology, and their electrochemical activity against nitrofurantoin (NF) was reported. The physico-chemical nature of LMNSs, and their texture and morphology were characterized by using X-ray photoelectron spectroscopy (XPS), Raman spectroscopy, powder X-ray diffraction (PXRD) and field emission scanning electron microscopy (FE-SEM) combined with energy-dispersive X-ray spectroscopy (EDX). The electrochemical studies were performed by using cyclic voltammetry (CV) and differential pulse voltammetry (DPV) techniques. The ensuing LMNSs have a fantastic active surplus area and uplifted surface volume ratio, which drives the selective and sensitive detection of NF. The as-synthesized LMNSs were coated on a screen-printed carbon electrode (SPCE) by a drop cast method, and the constructed LMNSs/SPCE acts as a potential sensing material towards the reduction of NF. Also, the designed sensor exhibits a broad linear range from 0.01 to 144 μM with a lower detection limit of 72 nM and sensitivity equivalent to 2.94 μA μM−1 cm−2 for the detection of NF. Meanwhile, the fashioned sensor has good selectivity in the presence of metal ions, biomolecules and similar nitro group containing drugs.


 Please wait while we load your content...
Please wait while we load your content...