A pre-formulation strategy for the liposome encapsulation of new thioctic acid conjugates for enhanced chemical stability and use as an efficient drug carrier for MPO-mediated atherosclerotic CVD treatment†
Abstract
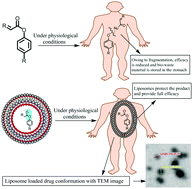
Lipoyl-apocynin and lipoyl-sesamol are bio-active conjugates of thioctic acid (also known as alpha-lipoic acid), synthesized using a benign chemical approach via the combination of thioctic acid and the powerful bio-phytonutrients, apocynin and sesamol, respectively. Encapsulation was performed for chemically modified thioctic acid conjugates within a liposome structure as a possible drug carrier for targeted site delivery without chemical degradation, which could be useful in myeloperoxidase (MPO) mediated atherosclerotic cardiovascular disease (CVD) treatment. A facile processing route for the thioctic acid conjugates with the complete chemical characterization and bio-encapsulation of these chemical entities in a liposome sphere was developed, and analysis to establish the encapsulation efficiency, zeta potential, vesicle size, surface morphology, and long-term sustained stability was performed. The liposome conjugates were also characterized via critical in vitro experiments. The encapsulation efficiency of the thioctic acid conjugates was found to be almost 70%. In addition, the vesicle size and zeta potential were also measured for the formulated thioctic acid conjugates. The chromatographic evaluation of the encapsulated conjugates revealed that the liposomes seem to protect the thioctic acid conjugates when incubated in simulated body fluid, preventing ester hydrolysis, thereby offering chemical stability to the thioctic acid conjugates. The liposome loaded conjugates were also determined to be potent agents for MPO mediated atherosclerotic CVD in relevant preclinical in vitro tests. This is the first report of a highly efficient and sustainable chemical method for the liposomal encapsulation of thioctic acid conjugates of apocynin and sesamol that has been substantiated using relevant in vitro assays, and it could be useful for MPO-mediated atherosclerotic CVD treatment.


 Please wait while we load your content...
Please wait while we load your content...