Nickel(ii) and nickel(0) complexes as precursors of nickel nanoparticles for the catalytic hydrogenation of benzonitrile†
Abstract
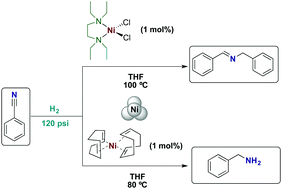
The use of the nickel(II) complex [(TEEDA)NiCl2] (1; TEEDA= N,N,N′,N′-tetraethyl-ethylendiamine) and nickel(0) complex [Ni(COD)2] (5) as pre-catalysts in the additive-free catalytic hydrogenation of benzonitrile (BN) is reported. In the presence of 1 (1 mol%), BN was hydrogenated under relatively mild reaction conditions (100 °C, 120 psi H2, 72 h) to the corresponding secondary imine, N-benzylidenebenzylamine (BBA), in very good yield (83%). As a counterpart, 5 (1 mol%) selectively hydrogenated BN to benzylamine (BA) in excellent yield (96%) under similar reaction conditions (80 °C, 120 psi H2, 24 h). In both cases, nickel nanoparticles (Ni-NPs) were identified as the catalytically active species. These Ni-NPs were formed in situ from 1 and 5 without external additives or additional stabilizers. The use of complex 5 was extended to the hydrogenation of different (hetero) aromatic and aliphatic nitriles.


 Please wait while we load your content...
Please wait while we load your content...