Sunlight assisted degradation of a pollutant dye in water by a WO3@g-C3N4 nanocomposite catalyst†
Abstract
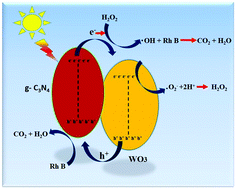
A series of WO3@g-C3N4 nanocomposites were synthesized and characterized by different physico-chemical techniques. The photocatalytic performance was investigated by degradation of Rhodamine B (100 ppm) in the presence of H2O2 under sunlight irradiation. The optimum loading of WO3 for the best performance is 2 wt%, at which the WO3@g-C3N4 nanocomposite exhibits the highest degradation efficiency of 99.5% under sunlight irradiation. The degradation with 2 wt% WO3@g-C3N4 follows pseudo first order kinetics and the rate constant is almost 2.6 and 7.7 times higher than that of ‘neat’ g-C3N4 and WO3. This WO3@g-C3N4 showed good recyclability with 94.2% efficiency after the seventh cycle. The enhancement of the photocatalytic efficiency of as-synthesized WO3@g-C3N4 nanocomposites may be due to the synergistic effect between WO3 and g-C3N4, high separation and easy transfer of photo-excited charge carriers at the interface of nanocomposites and the high specific area of the nanocomposites. The finding suggests that the as-synthesized WO3@g-C3N4 can exhibit high potential towards solar energy harvesting and environmental remediation.


 Please wait while we load your content...
Please wait while we load your content...