Remarkable improvement of TiO2 for dye photocatalytic degradation by a facile post-treatment
Abstract
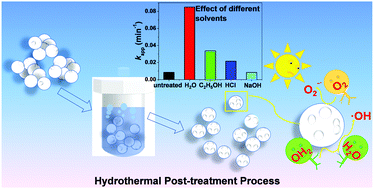
A simple hydrothermal post-treatment has been developed to construct a TiO2 photocatalyst with a desirable anatase phase, porous structure, large surface area, small particle size and hydrophilic surface. When aggregated nanoparticles are treated under a high temperature and pressure, the crystal structure of TiO2 is first deeply destroyed and then reconstructed. The dispersity can then be greatly improved and a typical mesoporous structure is simultaneously produced, accompanied with an increase in the specific surface area, average pore size, total pore volume and the production of oxygen vacancies. The structural changes have positive effects on the photocatalytic degradation of dye molecules. For example, the kinetic constant (0.085 min−1) can be increased by 10 times as compared with that for the untreated ones (0.0085 min−1). Other parameters including temperature, time, solvent, calcination treatment and type of TiO2 are also explored to optimize the treatment. In addition, electrochemical tests prove that the hydrothermal post-treatment for TiO2 results in an accelerated O2 reduction reaction and improved photo-induced charge transfer process. From radical quenching experiments, superoxide radicals (O2−˙) are proven to be the major oxidative species for both post-treated and untreated TiO2, but the production of hydroxyl radicals (˙OH) is dramatically promoted by the post-treatment, which is a key factor for the acceleration of the photocatalytic reaction.


 Please wait while we load your content...
Please wait while we load your content...