Low-temperature catalytic oxidation of benzene over nanocrystalline Cu–Mn composite oxides by facile sol–gel synthesis
Abstract
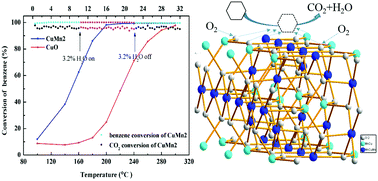
A series of nanocrystalline copper–manganese oxides (denoted as Cu3−xMnx, x = 0, 1, 1.5, 2, 2.5, 3, where x means the molar ratio of Cu and Mn) were successfully prepared by a facile citric acid sol–gel method. The combination of Cu2+ and Mn3+ is intensified and enhanced interface effects generated, which is beneficial for the catalytic oxidation of benzene. A series of analyses, such as X-Ray Diffraction (XRD), N2 adsorption–desorption, X-ray photoelectron spectroscopy (XPS), and hydrogen temperature programmed reduction (H2-TPR), were employed to further investigate the structural properties of the catalysts. An optimal Mn/Cu ratio of 2 forms CuMn2O4 spinels. CuMn2 with CuMn2O4 spinel structure presents a larger specific surface area, smaller pore diameter as well as more lattice oxygen species, exhibiting remarkable activity and stability for the catalytic oxidation of benzene. On account of these factors, CuMn2 possesses better low temperature reducibility and shows the best catalytic performance with 90% benzene conversion at 186 °C. The enhanced catalytic activity of CuMn2 is attributed to the stabilization of CuMn2O4 active phases and the intensive synergistic effect between Cu–Mn oxides. To prove the effect of CuMn2O4 spinel structure on catalytic performance, a CuO/Mn2O3 mixed catalyst (molar ratio 1 : 1) was prepared and applied to benzene oxidation (T90% = 198 °C), which indicates that the spinel structure has an encouraging effect on benzene catalysis. The catalytic properties of single copper oxide and manganese trioxide were also tested, the results show that CuMn2O4 has a crucial role in facilitating electronic transmission and mobility of the lattice oxygen.


 Please wait while we load your content...
Please wait while we load your content...