3-Amino-1-propanol and N-methylaminoethanol: coordination to zinc(ii) vs. decomposition to ammonia†
Abstract
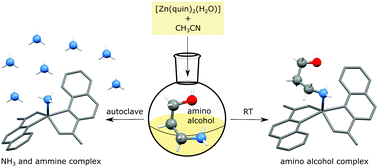
To broaden the limited knowledge concerning the zinc(II) coordination chemistry with amino alcohols, reactions of [Zn(quin)2(H2O)] (quin− = quinaldinate, C10H6NO2) with 3-amino-1-propanol (3-apOH, C3H9NO) and N-methylaminoethanol (N-maeOH, C3H9NO) were investigated. The starting material, zinc(II) with two quinaldinates coordinated in a bidentate chelating mode, provides a structurally rigid core with two sites available for interaction with amino alcohol ligands. When the reactions were carried out in acetonitrile in autoclaves at 105 °C, an unforeseen decomposition of amino alcohols to ammonia took place. This was accompanied by crystallization of an ammine complex [Zn(quin)2(NH3)] (1). Mass spectrometry of the gaseous phases confirmed unambiguously the presence of ammonia in such reaction mixtures. The desired complexes with coordinated amino alcohols could be obtained in good yield by carrying out the reactions at room temperature and/or in various solvents. Two novel amino alcohol complexes were prepared, [Zn(quin)2(3-apOH)] and [Zn(quin)2(N-maeOH)]. The 3-apOH ligand was coordinated to zinc(II) in a monodentate manner via the amino nitrogen. The 3-apOH complex crystallizes as an acetonitrile (2a), ethanol (2b), 2-propanol (2c) or water (2d) solvate. The conversion of 2a to 2d was monitored by IR spectroscopy. In [Zn(quin)2(N-maeOH)] (3), the N-maeOH ligand was coordinated in a bidentate chelating manner through both functional groups. DFT calculations were performed on the isomers of [Zn(quin)2(3-apOH)] and [Zn(quin)2(N-maeOH)], which differ in the binding modes of their amino alcohol ligands. Complete characterization of all compounds by means of X-ray structure analysis, infrared spectroscopy and NMR spectroscopy is presented.


 Please wait while we load your content...
Please wait while we load your content...