Highly ordered mesoporous flower-like NiO nanoparticles: synthesis, characterization and photocatalytic performance
Abstract
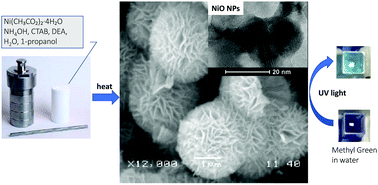
Highly ordered flower-like mesoporous NiO microspheres with a flower size of ∼2–5 μm and nanoflake size of ∼10–100 nm have been successfully synthesized via a chemical precipitation method assisted by hydrothermal treatment, using nickel acetate tetrahydrate as a metal source, ammonium hydroxide as a base, and diethanolamine (DEA) as a complexing agent. A Ni(OH)2 precursor was prepared with and without cetyltrimethylammonium bromide (CTAB) as a structure directing agent to investigate its effect on the size and morphology of the NiO particles. The physicochemical properties of the obtained NiO nanoparticles (NPs) were characterized using scanning electron microscopy (SEM), energy dispersive X-ray (EDX) spectroscopy, transmission electron microscopy (TEM), powder X-ray diffraction (PXRD), thermogravimetric and differential thermal analyses (TGA/DTA), Fourier-transform infrared (FT-IR) and diffuse reflectance ultraviolet visible (DR UV-vis) spectroscopies, and Brunauer–Emmett–Teller (BET) analysis. The results show that the obtained NiO NPs have a highly ordered nanostructure, with a moderate surface area of 21 m2 g−1, and a pore size of ∼6 nm. SEM, TGA/DTA and PXRD analysis indicate that CTAB is involved in the synthesis process and reduced the Ni(OH)2 size from 2 to 5 μm without having any significant effects on the particle morphology. The photocatalytic performance of NiO NPs was investigated under the illumination of UV light in the decolourization reaction of methyl green dye in aqueous solution. The NiO NPs exhibit excellent dispersion in the dye solution and considerable photocatalytic activity with a rate constant that is almost ten times higher than that of bulk NiO NPs.


 Please wait while we load your content...
Please wait while we load your content...