Studies on continuous selective hydrogenolysis of glycerol over supported Cu–Co bimetallic catalysts†
Abstract
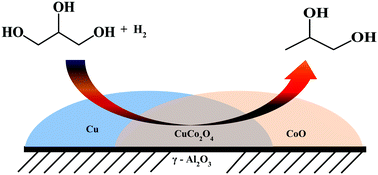
Alumina supported copper–cobalt catalysts were made and screened for continuous hydrogenolysis of glycerol to 1,2-propanediol at atmospheric pressure. BET surface area, temperature-programmed reduction, X-ray diffraction, pulse N2O chemisorption, transmission electron microscopy and X-ray photoelectron spectroscopy techniques were used to derive the characteristics of the catalysts. The presence of Co in Cu/alumina significantly increased the reducibility of CuO species and also the metallic Cu surface area. The catalyst with 10%Cu–7%Co on Al2O3 afforded complete glycerol conversion with 77% selectivity to 1,2-propanediol. The catalysts activity was found to be mainly due to the existence of Cu in a highly dispersed state with a high metal surface area and synergistic interactions between the Cu–Co moieties. The influence of the reaction parameters was investigated and the best possible parameters were determined. The most active catalyst showed high stability during the time on stream analysis.


 Please wait while we load your content...
Please wait while we load your content...