Functionalization of commercial pigment Hostasol Red GG for incorporation into organic thin-film transistors†
Abstract
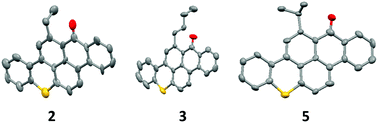
In the development of organic electronic devices, it is important to consider the overall cost of the carbon-based semiconductors. Many of the state-of-the-art semiconductors available today are prohibitively expensive, limiting their commercial functionality. The dye and pigment industries offer a wealth of cheap conjugated starting materials. In this study, we explored functionalizing the dye 14H-anthra[2,1,9-m,n,a]thioxanthen-14-one (Hostasol Red GG) through a simple one-pot Grignard reaction. Phenyl, primary and secondary alkyl substituents were introduced onto the parent thioxanthene benzanthrone, with 1,4-addition occurring preferentially to addition at the carbonyl for all products. The substituted Red GG derivatives were incorporated into organic thin film transistors. While the alkyl derivatives resulted in decreased device performance compared to native Red GG, transistor performance improved for the phenyl derivative. A combination of inexpensive starting materials and simple chemistry offers an abundance of possible new semiconductor targets for organic electronic devices with commercial potential.


 Please wait while we load your content...
Please wait while we load your content...