Large-scale surfactant-free synthesis of WS2 nanosheets: an investigation into the detailed reaction chemistry of colloidal precipitation and their application as an anode material for lithium-ion and sodium-ion batteries†
Abstract
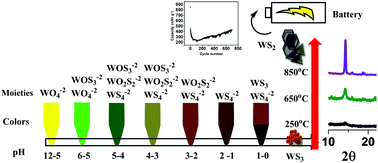
Transition metal dichalcogenides (TMDCs) are versatile 2D materials that have many important applications including their use as battery materials for energy storage. However, very few reports are available in the open literature on the scalable synthesis of these materials. In this work, an easy and versatile wet synthesis method for large scale preparation of tungsten disulfide (WS2) is reported. Although such methods are sometimes referred to in textbooks as the ‘industrial methods’ for WS2 manufacturing, a detailed synthesis process is not reported in the literature. The chemistry of the process, as revealed in this work, is also very different from that hitherto known. In this work, we clearly show that the sulfurization of WO42− is found to occur through several stable intermediates such as WOS32− and WO2S22−. Sulfurization is also found to occur through H2S upon lowering the pH. The entire process needs very careful monitoring as various side reactions and the corresponding pH are identified during the sulfurization process, which is crucial for product purity and yield. Apart from the sulfurization process, we also thoroughly explored the calcination step and hence have provided a complete process for obtaining the best pristine quality, scalable WS2 sheets. When evaluated as an anode material for Li-ion (LIB) and Na-ion (NIB) batteries, an initial capacity of about 400 mA h g−1 at 1C rate is obtained. A drastic capacity fading due to polysulphide formation is found to be more critical in the NIB system. The rate capability of pristine, bulk, scalable WS2 in LIBs is quite high (250 mA h g−1) even at 5C rate (2.1 A g−1). The initial results on carbon-coated WS2/C as the anode in LIBs show remarkable capacity recovery and retention, regaining the initial reversible capacity of about 450 mA h g−1 after 700 cycles at 1C.


 Please wait while we load your content...
Please wait while we load your content...