Complexation and fluorescence behavior of proflavin with chemically engineered amine capped carbon nanodots and its subsequent release into DNA environments†
Abstract
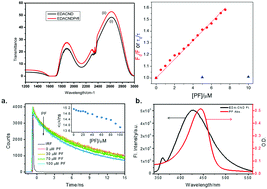
Carbon nanodots (CNDs) are one kind of fluorescent carbon nanomaterials, composed of polyaromatic carbon domains surrounded by amorphous carbon frames, and they have attracted a great deal of attention because of their interesting properties. They are a sort of fluorescent nanomaterial having unique optical properties as compared to other traditional organic fluorophores, which include high photostability and quantum yield. In our present study, CNDs and proflavin (PF) have been used as constituents of a drug delivery carrier, where PF is a very good DNA intercalator. Therefore, we hypothesize that DNA can be used as a sequestering agent for PF from a CND–PF nano-assembly. Amine capped carbon dots with a particle diameter of 80–100 nm are prepared by pyrolysis of citric acid. Probable excited-state interactions like fluorescence resonance energy transfer (FRET), photo-induced electron transfer (PET) etc. between PF and CNDs have been studied using several spectroscopic techniques e.g., steady-state absorption as well as fluorescence spectroscopy and time-correlated-single-photon counting (TCSPC) to measure fluorescence decay kinetics in the nanosecond–picosecond (ns–ps) time regime. The fluorescence intensity of the CNDs has been quenched by PF. After addition of CT DNA the quenched fluorescence intensity of the CNDs has been seen to be recovered. This indicates controlled release of PF into the DNA micro environment by CNDs, which shows their utility as an efficient drug delivery agent. Additionally, we have monitored the partial release of PF from the aforementioned complex into our selected “hydrophobic sink” of different compositions of a water–PEG-400 mixture.


 Please wait while we load your content...
Please wait while we load your content...