Good or evil: what is the role of water in crystallization of organometal halide perovskites?
Abstract
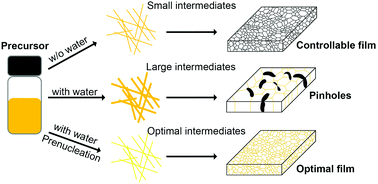
Perovskite solar cells (PSCs) have the potential to become one of the most cost-efficient photovoltaic devices. However, current fabrication methods of PSCs still require strict environment control and ultrahigh purity chemicals, which could prevent their large-scale commercialization. To tackle this challenge, the role of water is the first to be thoroughly understood in a perovskite formation process. Until now, there is still controversy about whether water is harmful or beneficial for perovskite formation, not to mention exactly what role water plays therein. In this Focus article, we review recent studies on water involved chemical reactions, solvent interaction, intermediates, and crystal growth in the perovskite film formation process, in order to bring out a full picture about what water does in the perovskite formation process. As our current understanding stands, a suitable amount of water could be of help for growing high quality perovskite films due to the resultant formation of intermediates, such as MAPbI3·H2O, which facilitates the conversion from precursors to perovskites. However, too much water would induce the formation of relatively stable components, such as (MA)4PbI6·2H2O, which are left in the product-films as impurities resulting in degraded device performance. Continual efforts should be made to further understand and develop water-involved strategies for consistent PSC fabrication under ambient conditions.
- This article is part of the themed collection: Focus article collection


 Please wait while we load your content...
Please wait while we load your content...