Tuning the electronic energy level of covalent organic frameworks for crafting high-rate Na-ion battery anode†
Abstract
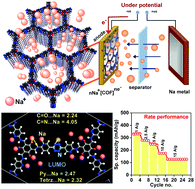
Crystalline Covalent Organic Frameworks (COFs) possess ordered accessible nano-channels. When these channels are decorated with redox-active functional groups, they can serve as the anode in metal ion batteries (LIB and SIB). Though sodium's superior relative abundance makes it a better choice over lithium, the energetically unfavourable intercalation of the larger sodium ion makes it incompatible with the commercial graphite anodes used in Li-ion batteries. Also, their sluggish movement inside the electrodes restricts the fast sodiation of SIB. Creating an electronic driving force at the electrodes via chemical manipulation can be a versatile approach to overcome this issue. Herein, we present anodes for SIB drawn on three isostructural COFs with nearly the same Highest Occupied Molecular Orbitals (HOMO) levels but with varying Lowest Unoccupied Molecular Orbitals (LUMO) energy levels. This variation in the LUMO levels has been deliberately obtained by the inclusion of electron-deficient centers (phenyl vs. tetrazine vs. bispyridine-tetrazine) substituents into the modules that make up the COF. With the reduction in the cell-potential, the electrons accumulate in the anti-bonding LUMO. Now, these electron-dosed LUMO levels become efficient anodes for attracting the otherwise sluggish sodium ions from the electrolyte. Also, the intrinsic porosity of the COF favors the lodging and diffusion of the Na+ ions. Cells made with these COFs achieve a high specific capacity (energy density) and rate performance (rapid charging–discharging), something that is not as easy for Na+ compared to the much smaller sized Li+. The bispyridine-tetrazine COF with the lowest LUMO energy shows a specific capacity of 340 mA h g−1 at 1 A g−1 and 128 mA h g−1 at a high current density of 15 A g−1. Only a 24% drop appears on increasing the current density from 0.1 to 1 A g−1, which is the lowest among all the top-performing COF derived Na-ion battery anodes.
- This article is part of the themed collections: Nanoscale Horizons 10th anniversary regional spotlight collection: Asia-Pacific and Nanoscale Horizons Most Popular 2020 Articles


 Please wait while we load your content...
Please wait while we load your content...